I am currently at La Selva biological research station collecting bats for a transcriptomic analysis. The purpose of this post is to record what we did for specimen preparation for future reference:
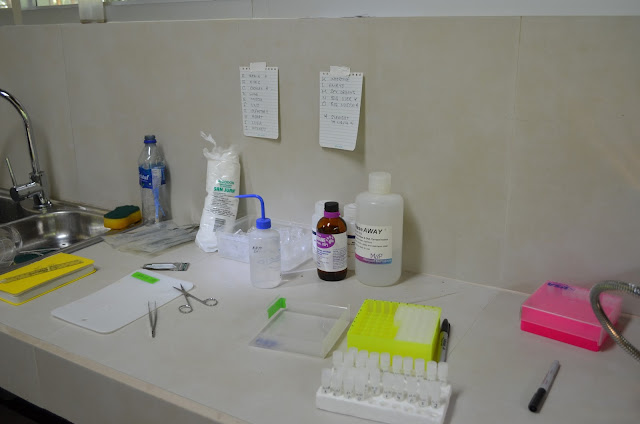
Lab set up for dissections of tissues:
The most organized way we found to do this was to perform the actions in the following order
To prepare the final specimen:
Lab set up for dissections of tissues:
Checklist:
- isofluorane (anesthetic)
- cotton balls
- RNAlater (lots of bottles!)
- RNAase AWAY
- cryovials (lots!)
- plastic droppers (lots!)
- boxes
- disposable vial rack
- sharpies (small and large)
- ethanol (to clean)
- bleach (to clean)
- plastic petri dishes for dissection
- dissection kit (I especially used sharp and dull forceps, scissors, small scissors, curved forceps)
- dissection scope
- cutting board
- light source for scope
- camera and lens
- tape
- organized binder with data sheets
- calipers/ruler and pesolas for measurements
- sponges
- bottles for sharps
- plastic squeezy bottles for RNA later
- Cooler (for ice)
- Little bucket for ice
- Ice
- Cooler (for dry ice if you want to collect and preserve the whole brain)
- Dry Ice (for brain)
- Wing punches
- scalpels
- liquid nitrogen and dry shipper
- tiny plastic baggies to put cryovials in
- string to hang bat bags on
- RNA protect
- DNA away
- ALL protect
- aluminum foil
Next time, I will get a voice recorder to record observations during dissection (which will also be useful in the field) and transcribe the notes later on.
- Make sheet before beginning with labeled tissues in respective order
- Bleach entire bench and wash down to prevent bleach from contaminating the tissue
- Wash everything with RNAase AWAY
- Final solution on top of everything with RNAlater
- Label cryovials
- Remove bat from bat bag
- Euthanize with anesthetic
- Immediately take measurements and record on data sheet before rigor mortis sets in
- Place cotton ball in mouth to prevent rigor mortis
- Separate head from body and have two people working on the dissection simultaneously to remove the different tissues
- Place tissues in respective labeled tubes
- Top off the tubes with RNAlater
- Place tubes on ice as soon as tissue is placed in RNAlater
- If harvesting the brain, gently scoop the whole brain into a pocket of powdery dry ice and cover with dry ice until the brain hardens. Be careful to not damage the olfactory bulb and keep in mind the strong optic nerve.
- Remove brain from dry ice. We had been placing it in RNA later but not sure if this is the best way to preserve the actual brain.
- Place all cryovials in tiny plastic baggies and label. Place the entire plastic bag in dry shipper.
- Place the remaining specimen in the foil to be frozen at -20 and later prepared in formadehyde
- Wash and clean bench in between each specimen.
The most organized way we found to do this was to perform the actions in the following order
To prepare the final specimen:
- Plastic sandwich baggies
- Bottles of formaldehyde
- WriteInTheRain paper (will not degrade in formaldehyde)
- Sharpies
- Gloves
- Masks

No comments:
Post a Comment